Latent and Active Failures Perfectly Align to Allow a Preventable Adverse Event to Reach a Patient
Problem: A physician prescribed 2 g of intravenous (IV) magnesium sulfate for a patient in palliative care to treat hypomagnesemia. To administer the dose, a night nurse went to an automated dispensing cabinet (ADC) and removed what was thought to be two 100 mL bags of magnesium sulfate 1 g, then hung both bags on the patient’s IV pole. The nurse attached the first 100 mL bag, scanned the barcode on the bag, and administered the infusion. After the 1 g magnesium dose had been administered, the nurse replaced the empty bag with the one remaining on the IV pole, but scanned the empty bag that had already infused to document the administration of the second bag. The nurse did not realize that due to a pharmacy error when refilling the ADC, the second bag contained midazolam 100 mg in 100 mL and administered that instead of magnesium. The patient experienced respiratory depression which the medical team initially attributed to progression of the patient’s illness. The patient had a “do not resuscitate (DNR)” order and thus aggressive measures including intubation were not to be used. Later, the nurse removed both empty bags from the IV pole and discovered that one of the bags was midazolam. The prescriber ordered several doses of flumazenil. Although the patient died later that morning, the erroneous administration of IV midazolam was not considered to be a proximal cause of death.
Most preventable adverse events, including this one, happen when multiple latent failures in the organization align perfectly with the active failures of individuals. Latent failures refer to less apparent failures embedded in the organizational systems of care, the environment, or equipment, which often go unrecognized until they harm patients. Organizational latent failures (e.g., lack of, inaccurate, or incomplete policy or procedure) are less obvious than the active failures of individuals, such as human error (e.g., misprogramming a pump) or at-risk behaviors (e.g., choosing not to follow a procedure). Thus, latent failures are “accidents waiting to happen”—they often make it easier for an individual to make an error or engage in an at-risk behavior. It is the job of leaders at all levels within healthcare to identify and address latent failures that exist upstream before errors have a chance to reach our patients.
Many are familiar with James Reason’s “Swiss cheese” model used to describe how latent and active failures lead to preventable adverse events. Reason suggests that a system is analogous to a stack of Swiss cheese slices. Each slice represents a part of the organizational system that defends against errors. A hole or gap in one slice of cheese, or system, represents a latent failure that may allow an active failure to get through a single layer. But in the subsequent layers, if the holes are not aligned, the error may be prevented before it reaches a patient. For a preventable adverse event to occur, the latent failures (holes in the cheese) need to align perfectly with the active failures of individuals to get through the many defense layers of the system and reach the patient. As you read additional details about the event described below, notice how a series of latent and active failures can be identified at multiple steps in the medication-use process.
Similar bag labels inside the overwrap (latent failure)
Both midazolam and magnesium sulfate premixed products, manufactured by WG Critical Care, are in 100 mL bags within aluminum overwraps. The label on the overwraps has features to differentiate the products, including the addition of a high-alert medication symbol and a bright orange rectangle containing the drug name on the midazolam overwrap (Figure 1). However, once practitioners remove the IV bags from the overwrap, the inner labels printed on the bags are not as easy to distinguish and may contribute to look-alike errors (Figure 2).
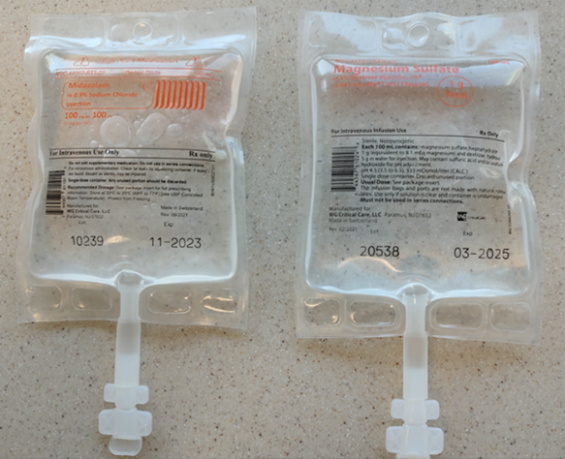
Unaccounted for midazolam discrepancy (active failure)
A few days before this event, a pharmacist reported a single midazolam bag discrepancy in the controlled substance safe. After a pharmacy supervisor could not account for the missing bag, the pharmacy cleared the discrepancy in the safe. However, the root cause of the discrepancy was never determined.
Scanning process flaw (latent failure)
Although the pharmacy required technicians to scan medication barcodes when refilling the ADC, the process (as designed) only prompts scanning of the first barcode when refilling multiple units of the same medication (e.g., scanning the barcode on one bag to open the bin, and then refilling all the required bags without scanning each one).
Stocking error (active failure)
The organization’s investigation revealed that a pharmacy technician had misplaced the midazolam bag in the magnesium bin in the ADC during the filling process.
Selection of the wrong infusion (active failure)
Although the nurse looked at the labels of the two infusions that had been removed from the ADC, the difference between the two medications was not noticed.
Difficulty scanning the barcode (latent failure)
Infusion bag barcodes have been chronically challenging for practitioners to scan.
Proxy scan (active failure)
Because some barcodes on infusion bags were difficult to scan, nurses had developed a workaround (at-risk behavior), where they scanned the barcode on the empty infusion bag already hanging instead of the subsequent infusion bag, believing it to be the same infusion. This allowed the nurse to document the subsequent infusion quickly but, in this case, inaccurately.
Low lighting (latent failure)
The adverse event occurred during the night shift, where there was deliberate low-level lighting in the patient’s room. Thus, the nurse had difficulty reading the labels of the bags hung on the IV pole.
Safe Practice Recommendations: This event demonstrates that, typically, many things must go wrong for a medication error to reach the patient. To minimize errors, identify active and latent failures and evaluate your processes by considering the following recommendations:
New product review. When the pharmacy receives a new product (e.g., new product added to formulary, drug shortage), conduct a review to identify potential risks with the product’s design and/or packaging including any issues with scanning the product’s barcode. Also, identify any look-alike labeling and packaging concerns with other products on the formulary. Communicate similarities with manufacturers, ISMP, the US Food and Drug Administration (FDA), purchasers, and group purchasing organizations as appropriate.
Purchase from a different manufacturer. When problems are recognized, consider purchasing the product (or one product of a problematic pair) from a different manufacturer. We reached out to the manufacturer, WG Critical Care, to notify them of this event and to recommend differentiating the infusion bag labels.
Prompt resolution of discrepancies. A controlled substance discrepancy could be an indication that the pharmacy dispensed the wrong product or there could be a diversion issue. Practitioners may be able to identify a dispensing or stocking error prior to it reaching a patient. Investigate and identify the root cause of discrepancies and educate practitioners to escalate concerns to leadership for prompt resolution, which may involve designating resources (e.g., pharmacy staff) to physically check the stock in the pharmacy and in all ADCs.
Manage pharmacy ADC stock. Designate an area in the pharmacy for ADC stock management with space to avoid the intermingling of medications and minimal interruptions and distractions.
Employ dispensing barcoding technology. Use barcode scanning technology in the pharmacy to confirm that medications chosen for distribution to the ADC match the medications listed on the ADC fill report. Segregate and secure all medications designated for an individual ADC during transport. Use barcode scanning at the cabinet to promote the accurate placement of medications in the correct drawer or pocket location. Determine if your ADC has the functionality for practitioners to scan each individual product when refilling the ADC, and consider requiring barcode scanning of each medication before placing it in the ADC. Review the ISMP Guidelines for the Safe Use of Automated Dispensing Cabinets (Core Safety Process #6).
Require an independent double check during selection. Provide a final independent double check of all medications selected in pharmacy for ADC distribution to ensure the right drug, strength, dosage, and correct quantity are verified. Even if barcode technology is used in the selection process, a physical independent double check should be done in the pharmacy prior to distribution.
Promote optimal conditions. Ensure the physical environment offers adequate space and lighting and allows practitioners to remain focused on the medication-use process (e.g., drug selection, barcode scanning) without distractions.
Read overwrap and inner labels. Carefully review individual product labels after removing the medication from the ADC, when removing infusions from overwraps, when spiking an IV bag, and prior to administration.
Employ bedside barcode technology. Use bedside barcode scanning technology to confirm that medications selected for administration match the patient’s medication administration record. Coach staff to never use a proxy scan, such as scanning the barcode on an already hanging empty bag or a medication label not affixed to what is actually being administered.
Troubleshoot difficult barcodes. Test new product barcodes in the pharmacy prior to distribution. If a practitioner has trouble scanning a barcode, manufacturers have suggested holding the scanner 4 to 6 inches from the bag, scanning at an angle, and/or putting a contrasting color behind the bag. Develop a process within the organization for end users to report barcode issues so that pharmacy leadership can consider an alternative product, when possible. Report barcode scanning issues to ISMP so we can work with manufacturers and FDA to improve the safety of product labeling and packaging. Instruct staff on the accepted best practice to use when a barcode scan does not work.
Summary. We encourage practitioners who investigate events to always consider multiple latent system failures and multiple active failures by practitioners that might have contributed to the error or hazard. Our natural tendency is to look for simple, singular answers during event investigations, and these often focus on errors at the sharp end—the active failures. But there are often many hidden twists and turns along the path to a medication error. By themselves, latent failures are often subtle and may not cause problems. Their consequences are hidden, becoming apparent only when they occur in proper sequence and are combined with the active failures of multiple individuals to penetrate or bypass system safety nets. This event provides clear evidence that medication errors are almost never caused by the failure of a single system or the fault of a single practitioner. Rather, a preventable adverse event like this is the result of the combined effects of latent failures in the system and active failures by practitioners. Therefore, the goal of the investigation should be to proactively make system changes to correct latent failures, making it harder for an active failure by a practitioner to result in an error reaching a patient.
Suggested citation:
Institute for Safe Medication Practices (ISMP). Latent and active failures perfectly align to allow a preventable adverse event to reach a patient. ISMP Medication Safety Alert! Acute Care. 2023;28(1):1-4.
Access this Free Resource
You must be logged in to view and download this document.
