Multiple Error Pathways with the Monoclonal Antibodies, Casirivimab and Imdevimab
Problem: On November 21, 2020, the monoclonal antibodies, casirivimab and imdevimab (REGEN-COV), received initial Emergency Use Authorization (EUA) from the US Food and Drug Administration (FDA). The two monoclonal antibodies are authorized to be administered together for the treatment of mild to moderate coronavirus disease 2019 (COVID-19) in adults and children (12 years and older weighing at least 40 kg) with positive results of direct, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) viral testing, and who are at high risk for progression to severe COVID-19, including hospitalization or death. The monoclonal antibodies are not authorized for use in patients who are hospitalized due to COVID-19, require oxygen therapy, or an increase in oxygen therapy if already receiving chronic oxygen therapy.
The EUA has been revised several times to address new safety information and to allow use for post-exposure prophylaxis of COVID-19 in adults and children (12 years and older and weighing at least 40 kg) who are at high risk for progression to severe COVID-19, including hospitalization or death, and are not fully vaccinated or are not expected to mount an adequate immune response to vaccination. The EUA also allows post-exposure prophylaxis in individuals at high risk of exposure to another person infected with SARS-CoV-2 in the same institutional setting (e.g., nursing homes, prisons). Under the current EUA, the monoclonal antibodies can be administered either subcutaneously or intravenously (IV). The IV route is strongly preferred for treatment; however, either route is recommended for post-exposure prophylaxis. The subcutaneous route takes less time, facilitating administration, such as by pharmacists, and allowing for more widespread availability.
In a September 23, 2021 SAFETY brief, we mentioned possible confusion with a new alternative packaging for casirivimab and imdevimab. Due to high demand for the monoclonal antibodies, Regeneron (manufacturer of REGEN-COV) is distributing co-packaged cartons of the antibodies that are manufactured by Regeneron’s development partner, Roche Pharmaceuticals. The Roche co-packaged products are intended for distribution outside the US and use labeling that differs in some ways from Regeneron’s other REGEN-COV products. In the SAFETY brief, we warned about possible confusion with the foreign Roche label because it lacks a National Drug Code (NDC) number, the product is labeled “For Pandemic Use” instead of “For EUA Use,” and a barcode may not be present or functional. We also warned about confusion among the various presentations of the monoclonal antibodies.
Available Product Presentations
Currently, casirivimab and imdevimab are available in the US in a co-formulated vial containing both casirivimab and imdevimab together in the vial, co-packaged in cartons containing one vial of casirivimab and one vial of imdevimab, and in Dose Pack bags containing individual vials of casirivimab and imdevimab (Table 1).
Table 1. Casirivimab and imdevimab packaging presentations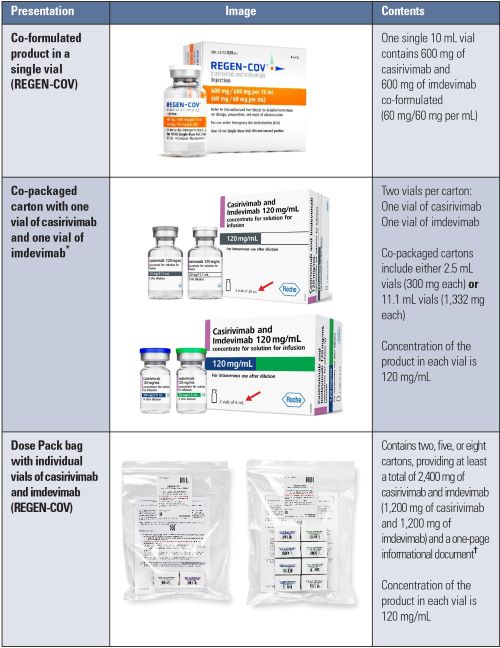
* The co-packaged cartons are manufactured by Regeneron’s development partner Roche Pharmaceuticals and are being distributed by Regeneron to increase the availability of doses of casirivimab and imdevimab.
† The included one-page informational document contains inaccurate dosing information per the current Health Care Provider Fact Sheet. The QR code on the document can be used to obtain the most current Fact Sheet.
Risk of Errors
We are hearing that many nurses have been tasked with selecting these monoclonal antibodies from automated dispensing cabinets (ADCs) and then preparing and administering them. Furthermore, the locations where casirivimab and imdevimab are administered (e.g., infusion centers, clinics, emergency departments, long-term care facilities, prisons) are extremely busy, and the staff are often rushed, increasing the risk of an error. Under these conditions, we have received numerous reports of confusion as well as actual errors. Most of the errors are associated with preparing and administering only one component of the two monoclonal antibodies, or prescribing, preparing, and/or administering the wrong dose. Common causes of this confusion and examples of the error reports we have received just in the past few weeks are summarized below.
Common Causes of Confusion and Errors
Prescribing errors. The monoclonal antibodies have been prescribed using an ambiguous dose designation of “600 mg” instead of the recommended “600 mg of casirivimab and 600 mg of imdevimab” (or “300 mg of casirivimab and 300 mg of imdevimab” for certain post-exposure prophylaxis circumstances). A common contributing factor associated with these prescribing errors is misleading order entry fields that do not make it clear that 600 mg (or 300 mg) of EACH monoclonal antibody is required for appropriate dosing. Based on error reports we have received, a knowledge deficit about the recommended dose has infrequently been an underlying cause of prescribing errors.
Selection errors. Organizations may not have a choice of which product presentation they receive. Thus, given the various presentations of the monoclonal antibodies and the fact that they are often stored together, selection errors among the different presentations have been occurring in both the pharmacy and in patient care areas where the antibodies might be prepared. For example, mix-ups between vials of the co-formulated antibodies and vials of the individual antibodies have been reported.
Preparation errors. Some preparation errors have been related to label confusion. Displayed on the front panel of the Roche label for the co-packaged cartons containing 2.5 mL or 11.1 mL vials of casirivimab and imdevimab is “2 vials of 6 mL” or “2 vials of 20 mL,” respectively (see the red arrows on the images in Table 1), which refers to the vial size, not the contents of each vial or the sum of the two vial contents. Confusion about the volume in each vial could lead to preparation of incorrect doses. ISMP has received several reports regarding this particular label confusion. One reported error involved a pharmacy technician who withdrew the contents of both casirivimab and imdevimab from the Roche co-packaged 2.5 mL vials. Upon discovering that the total volume was only 5 mL instead of 6 mL (which was listed on the front label panel), the technician thought she did not have enough medication to prepare a 600 mg total dose (300 mg of each monoclonal antibody) for repeat dosing intended for post-exposure prophylaxis.
Confusion has also been reported regarding the total contents of the 10 mL vial of the co-formulated monoclonal antibodies. Some have mistakenly thought that the co-formulated product contains 300 mg of each antibody (600 mg total), when it actually contains 600 mg of each antibody (1,200 mg total). In one reported error, a nurse retrieved the co-formulated product and misunderstood “600 mg/600 mg per 10 mL” on the label to mean that each 10 mL vial contained 600 mg in total (300 mg of casirivimab and 300 mg of imdevimab). She then retrieved a second vial of the co-formulated product and mixed the two 10 mL vials in a 100 mL infusion bag of 0.9% sodium chloride injection, thus administering an overdose. Confusion has also been reported regarding whether each vial contains the co-formulated monoclonal antibodies (both antibodies) or contains a single antibody.
Errors related to the various instructions for preparation following Regeneron’s Fact Sheet have also been reported. For the recommended IV infusion route of administration, a 10 mL (600 mg/600 mg) vial of the co-formulated product, or 5 mL of casirivimab (600 mg) and 5 mL of imdevimab (600 mg) from individual antibody vials, must be injected into a 50 mL, 100 mL, 150 mL, or 250 mL, single 0.9% sodium chloride or 5% dextrose infusion bag, each with a different minimum infusion time and the 50 mL bag with a different rate of infusion. We have heard about nurses who have prepared each monoclonal antibody in a separate infusion bag, rather than preparing them together in a single bag. We also learned about a nurse who selected two 2.5 mL vials of imdevimab (each labeled 300 mg/2.5 mL) and prepared and administered them without the casirivimab component, misunderstanding the “600 mg” dose designation that was listed on the patient’s medication administration record.
The various presentations of these monoclonal antibodies, and the fact that some co-packaged dose packs contain enough product for two full doses instead of just one full dose, increases the risk of an error. Also, we are not confident that vials of the monoclonal antibodies that are stored under refrigeration are consistently being brought to room temperature as directed for 20 minutes prior to dilution or subcutaneous injection.
For administration by the subcutaneous route, different preparation directions are provided in the Fact Sheet. If using a casirivimab and imdevimab co-formulated 10 mL vial, four separate syringes (2.5 mL each) must be prepared for 600 mg of casirivimab and 600 mg of imdevimab (or two syringes for 300 mg doses of each antibody). If using casirivimab and imdevimab individual vials, two separate syringes (2.5 mL each) must be prepared for both casirivimab and imdevimab (for a total of four syringes) to provide a 600 mg dose of each antibody (or just one syringe of each for a total of two syringes for 300 mg doses of each antibody). While we have not received any reported errors regarding preparation of the monoclonal antibodies for the subcutaneous route of administration, we worry that errors might be occurring, and the syringes might not be labeled during preparation in patient care areas.
Administration errors. Most of the reported administration errors we received have been noted above (e.g., wrong doses, administering just one monoclonal antibody instead of both, wrong rates of infusion [dependent on bag size]). We have also received reports where the four syringes prepared for subcutaneous administration were administered at the same injection site instead of different injection sites as recommended. It is also unclear how the most up-to-date information in the current casirivimab and imdevimab Fact Sheet is being conveyed to frontline practitioners who have to prepare and administer the monoclonal antibodies, particularly since the Fact Sheet has been revised multiple times.
Safe Practice Recommendations: To reduce the risk of errors when prescribing, preparing, dispensing, or administering casirivimab and imdevimab, take the following precautions:
Clarify dosing during order entry. In order entry systems and on standardized order sets, make it clear that 600 mg (or 300 mg under certain conditions) of each antibody is required for appropriate dosing. Consider requiring the entry of an indication (e.g., treatment, post-exposure prophylaxis, repeat dose of post-exposure prophylaxis) and default to the appropriate dose based on the indication.
Require pharmacy preparation. Whenever possible, outside of emergencies, have the pharmacy prepare and label patient-specific subcutaneous doses and IV infusions of the monoclonal antibodies, and dispense them to the appropriate patient care areas. If practitioners must prepare the doses in patient care areas, provide them with clear preparation instructions and preprinted labels for the subcutaneous syringes and the IV infusions.
Update EHRs with current information from the Fact Sheet. While it is not easy, for all EUA drugs, including casirivimab and imdevimab, it is important to assign an individual to regularly check the Fact Sheets for the most current information and to keep all electronic health record (EHR) systems updated with new information so it is readily available to all frontline practitioners. If questions arise, it is equally important for all practitioners prescribing, preparing, dispensing, and administering casirivimab and imdevimab to refer to the current Fact Sheet in case it has been revised. Revision dates and recent major changes are described within the first few pages of the Fact Sheet. Do not use the dosing and administration information on the one-page document in the Dose Packs because this information is not current.
Create separate storage. Separate the different presentations of the monoclonal antibodies in sequestered storage containers in the refrigerator and other storage locations, and clearly label each storage container. Consider packaging each complete dose in a separate, labeled ziplock bag.
Use auxiliary warnings. Include bold, colorful critical warnings on the product storage containers and on electronic screens or menus where these products are listed, based on the type of errors that have been reported. At a minimum, consider using these auxiliary warnings: “Co-formulated casirivimab and imdevimab,” “Co-packaged casirivimab and imdevimab (must be administered together),” and “Dose Pack of casirivimab and imdevimab (must be administered together).”
Reduce confusion with the Roche co-packaged product. If you receive the Roche co-packaged monoclonal antibodies, educate staff about the label differences. Also, before product use, either place (if absent) or replace the barcode on the product with a pharmacy-prepared barcode, or test any available barcodes on the product and manually input the product information, including the NDC number, into your EHR system to ensure the barcodes do not provide incorrect information when the product is scanned. If questions arise, ensure staff know to reference the Fact Sheet intended for US administration rather than the package leaflet in the carton (which is not approved for use in the US and should be discarded). Also consider clarifying the volume of product contained in each vial using auxiliary labels.
Educate staff. Educate practitioners who prepare and administer the monoclonal antibodies, with a particular focus on the various presentations available, reported label confusion, the types of errors reported nationwide, and preparation and/or administration instructions from the most current Fact Sheet.
Access this Free Resource
You must be logged in to view and download this document.
