FDA Guidance Needed to Assure Safe Labeling Practices by 503A and 503B Compounders
Problem: Increasingly, ISMP has been receiving complaints and reports of errors, some serious, about prefilled compounded syringes that look alike. Drug shortages may be worsening the situation, as hospitals may be relying more on compounders they have not previously used to fulfill their needs. The US Food and Drug Administration (FDA) does not hold the labeling of drugs compounded or repackaged by pharmacies or outsourcing facilities to all of the same standards as FDA-approved products. For example, if certain conditions are met, federal law exempts compounded drugs from the requirement for labeling with adequate directions for use. Further, ISMP has become aware that some compounders deviate from USP <7> labeling standards. Specifically, under USP <7>, the strength per total volume should be the primary and prominent expression on the principal display panel of the label, followed by the amount per mL enclosed by parentheses (a USP requirement since 2009). However, ISMP has observed that the strength per mL has often been used as the primary expression on compounders’ labels, leading to inconsistencies between products available in the hospital, thus creating unsafe conditions. Errors have occurred when the more prominent per mL strength is mistaken as the total amount of drug in the syringe. Such errors were the impetus for the USP <7> requirement for prominence of the strength per total volume on labels.
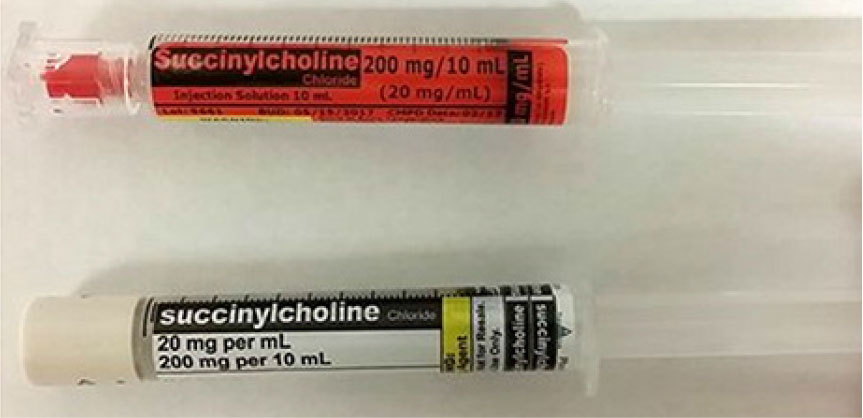
Our March 23, 2017, newsletter described a case in which a pharmacist and an anesthesiologist reported finding two syringes of succinylcholine with the strength displayed differently, both in the same pocket of the anesthesia cart in the operating room (OR) suite. Each syringe contained the same amount of drug, but one, from Cantrell Drug Company, was labeled using the USP <7> standard of strength per total volume, while the other one, from PharMEDium, listed the amount per mL as the primary display of concentration (Figure 1). Both practitioners thought a significant medication error could occur if the succinylcholine strength was misidentified. Apparently, syringes from the two compounders had been purchased and stocked without noticing and addressing the labeling inconsistency.
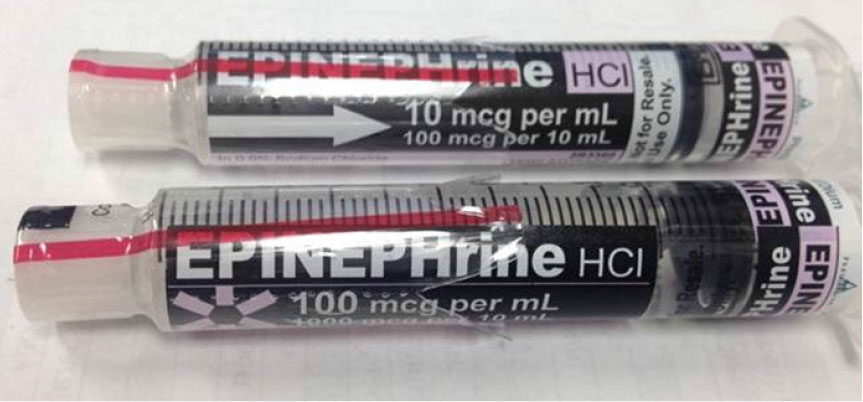
ISMP has also received reports of high-dose, 1 mg per 10 mL (100 mcg/mL) EPINEPHrine syringes that were placed in OR syringe bundles that normally contained 100 mcg per 10 mL (10 mcg/mL) EPINEPHrine syringes (Figure 2). To prevent look-alike product mix-ups, pharmacy staff had affixed auxiliary labels to the syringes to differentiate between the high- and low-dose EPINEPHrine. However, a technician misapplied a 10 mcg per mL auxiliary label to a high-dose syringe.
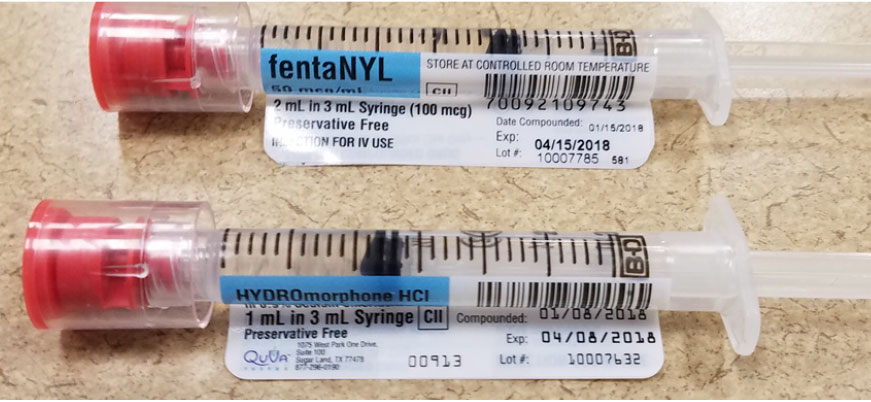
There has also been confusion reported between HYDROmorphone and fentaNYL syringes from QuVa Pharma, a 503B compounder. While the two syringes clearly communicate their respective drug names, there are other similarities that may draw attention away from reading and discerning the drug name. These similarities include using the same ASTM International standard blue color code for opioid analgesics on the label, a flagged white section of the label common to both syringes, the same syringe sizes, and same red color for the tamper-evident caps (Figure 3).
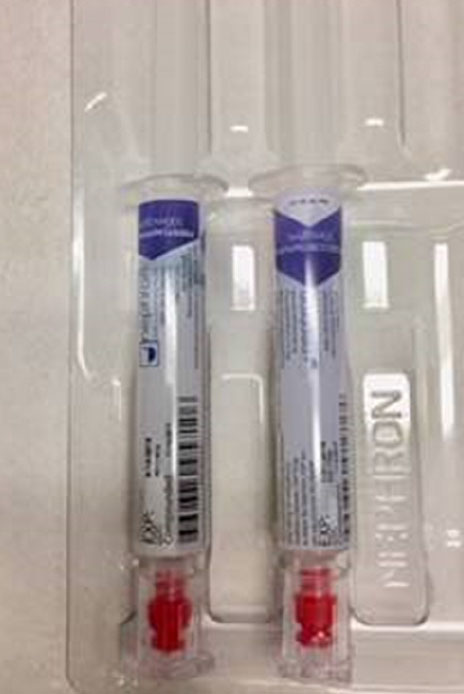
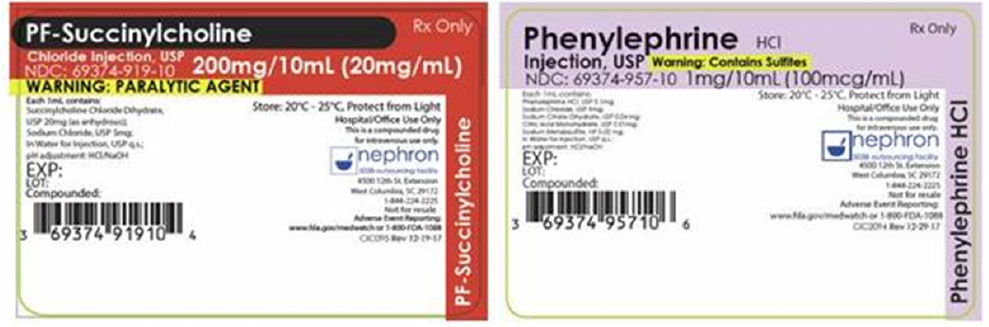
Another look-alike compounded syringe error reported recently involved a patient in a hypotensive state during surgery. FDA received an expedited manufacturer report from Nephron Pharmaceuticals about a patient given the neuromuscular blocking agent succinylcholine instead of the intended vasoconstrictor medication, phenylephrine. The patient’s hypotensive state was prolonged due to the medication error. Fortunately, the event resolved without further medical intervention as the patient was already intubated and under anesthesia at the time of the inadvertent administration of succinylcholine. The person reporting the incident mentioned that both syringes were “placed backwards” (drug name down) in the tray, with the Nephron logo facing up, making the syringes look similar. Figures 4a and 4b show the Nephron succinylcholine and phenylephrine syringes, with labels showing the drug names in one orientation (Figure 4a) but looking identical when placed drug name down in the tray (Figure 4b). The company recently revised these labels (Figure 4c).

Based on reports sent to ISMP and FDA, 503A pharmacies and 503B outsourcing facilities inconsistently follow label guidelines required of manufacturers under FDA and USP standards. Look-alike labeling is also a problem, and there are other serious issues that have contributed to errors. For example, some compounders package highly concentrated, high-alert medications in a single syringe, with the expectation that the entire contents or aliquots will be used to make diluted drug infusions. However, mishaps may occur that result in the administration of the entire syringe contents, undiluted.
Safe Practice Recommendations: FDA should not allow products from compounders to follow different container labeling standards than commercial manufacturers, thus creating unsafe conditions. Unfortunately, sections 503A and 503B of the Federal Food, Drug, and Cosmetic Act exempt compounded drugs from some of the labeling required of commercial drug manufacturers. FDA should convene a Pharmacy Compounding Advisory Committee meeting to review relevant error reports received since implementation of the Compounding Quality Act and sections 503A and 503B. The goal should be to publish a guidance that calls upon compounders to follow the same safety standards required of commercial manufacturers, including a requirement for 503B compounders, which are regulated by FDA under the Act, to submit their labels to FDA for review. While regulatory changes are considered, compounders should voluntarily comply with the same labeling standards currently required for commercial manufacturers.
