Do You Know What Doses Are Being Programmed in the OR? Make it an Expectation to Use Smart Infusion Pumps with DERS
Smart infusion pumps with dose error-reduction systems (DERS) have become the standard of care for parenteral medication delivery. These devices allow organizations to create a tailored library of medications and hydrating solutions with standard concentrations, dosing/infusion limits, and alerts (e.g., clinical advisories, soft stops, hard stops). Smart pumps with DERS can detect dosing and programming errors, and thus have been shown to reduce the incidence of medication errors.1-6 They can also provide a great deal of data that is useful in improving safe practices.
Use of smart pumps in the OR
The use of smart infusion pumps is most critical in patient care areas where high-alert medications are administered, including in the operating room (OR). While use of smart pumps in patient care locations outside the OR has spread,7,8 lower usage in the OR, endoscopy suites, and other procedural areas, particularly for epidural infusions, has been observed.8 Furthermore, compliance with engaging DERS, a vital decision support system, is lower in these procedural areas, risking potentially harmful errors.
To cite one example, a certified registered nurse anesthetist (CRNA) programmed a smart pump to deliver a dexmedetomidine infusion (4 mcg/mL) at a rate of 0.15 mcg/kg/minute instead of 0.15 mcg/kg/hour for several hours before the error was noticed. With the patient’s weight of 66 kg, this resulted in an infusion rate of 148 mL per hour instead of the correct 2.5 mL per hour. Instead of selecting dexmedetomidine from the drug library when programming the smart pump, the CRNA used an infusion mode outside the DERS and accidentally selected “Min” for minutes instead of hour. The pump did not issue a dose warning since the DERS had not been engaged. No long-term adverse effects were reported since the patient was closely monitored, and any short-term cardiovascular effects were quickly corrected during the surgical procedure.
Barriers to the use of smart pumps in the OR
Traditionally, there are several known barriers to optimal use of smart infusion pumps with DERS, including programming and workflow issues; pump technology limitations and usability issues; resource-intensive builds, maintenance, and updates of customized drug libraries; issues with secondary and intermittent infusions; problems with wireless connectivity, including security (potential hacking) issues; and DERS decision support challenges such as alert/alarm fatigue and the ease of opting out.8 In addition to these, unique barriers and challenges may exist in the OR:
-
Unclear or no expectation for anesthesia providers and other OR practitioners to use smart pumps with an engaged DERS
-
Not educating anesthesia providers and other OR practitioners to promote understanding of the capabilities and limitations of smart infusion pumps, including loading and bolus dose capabilities
-
Failure to understand that “anesthesia mode” settings (available with some smart pumps) reduce all hard stops to soft stops, thereby allowing overrides of dosing limits that had been established with the intent of never being bypassed
-
Different concentrations and dosing units between the OR and critical care areas to which postoperative patients may be transferred with medication infusions that were started in the OR
-
Complexity of the process for selecting the proper library when transferring patients from the OR to the post-anesthesia recovery unit and/or inpatient unit
-
Perception of unacceptable soft or hard dose/infusion rate limits for medications/hydrating solutions used in the OR
-
Frequency of loading and bolus doses administered in the OR
-
Not including anesthesia providers and other OR practitioners on the team when building the anesthesia/OR drug library
Notwithstanding these issues, one of the most fundamental ways to improve medication safety in the OR is to ensure anesthesia providers and other OR practitioners are employing smart infusion pumps with DERS.
Data analytics regarding excessive doses in the OR
Once anesthesia providers and other OR practitioners are using smart infusion pumps, the best way to provide visibility of the practices associated with medication administration in the OR is through evaluation of the pump data. Historically, this data has focused on compliance with engaging DERS and the management and reduction of alerts and alarms. While these measures are critically important, there is additional value in this data to understand the range of doses administered, including doses that could be supratherapeutic and harmful.
To this end, ISMP has reviewed data from the Bainbridge Health* infusion pump network to analyze the prevalence with which anesthetic or sedation agents are programmed in excess of the recommended dosing range. Below are just two examples from network hospitals that requested help evaluating the dosing patterns of commonly used medications administered by anesthesia providers in the OR.
Propofol dosing
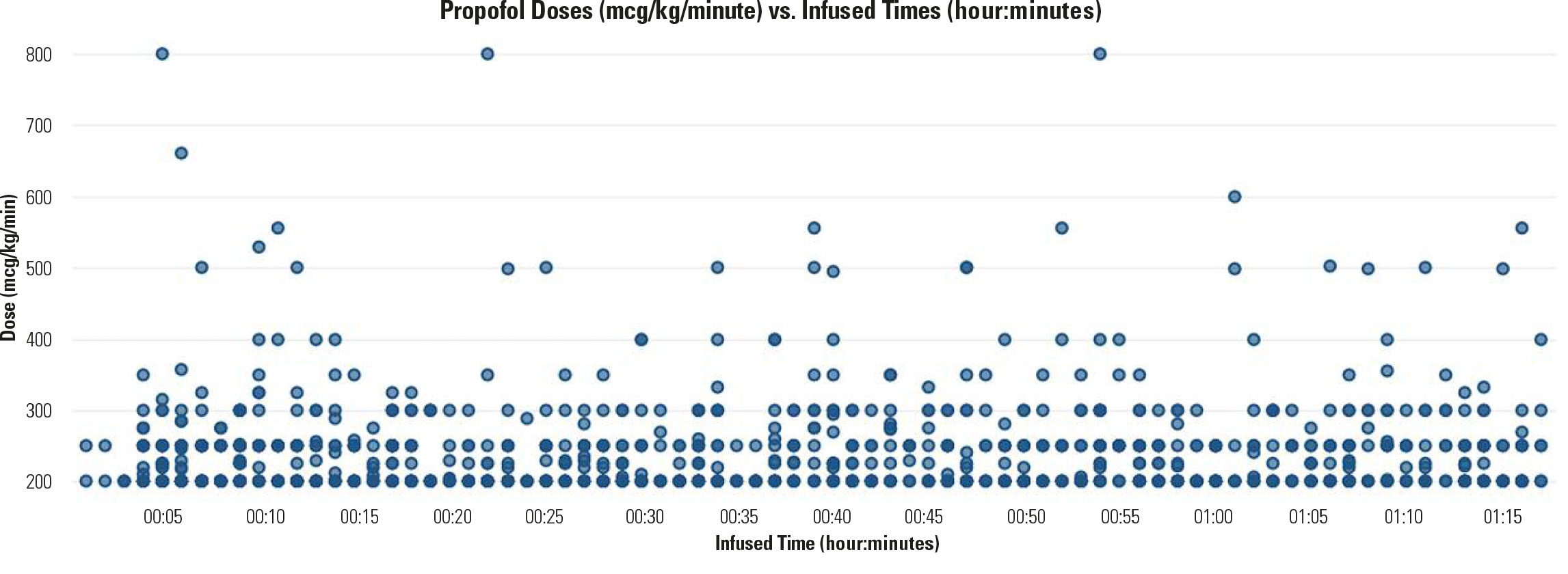
Propofol is a common general anesthetic agent administered during procedures. The usual adult dose of propofol for maintenance of general anesthesia is 100 to 200 mcg/kg/minute. Figure 1 shows the distribution of propofol doses administered at or above the upper adult dosing limit of 200 mcg/kg/minute in 16 network hospitals during the calendar year 2019. The figure shows that, over 12 months, many doses were programmed at or above 200 mcg/kg/minute. Even if the upper dosing limit was set at 300 mcg/kg/minute, as it is for pediatric patients, quite a few doses exceeded this amount. Doses were as high as 800 mcg/kg/minute, which infused for more than 50 minutes in one case. Propofol can cause significant patient harm at high doses, notably severe hypotension and respiratory depression.
Dexmedetomidine dosing
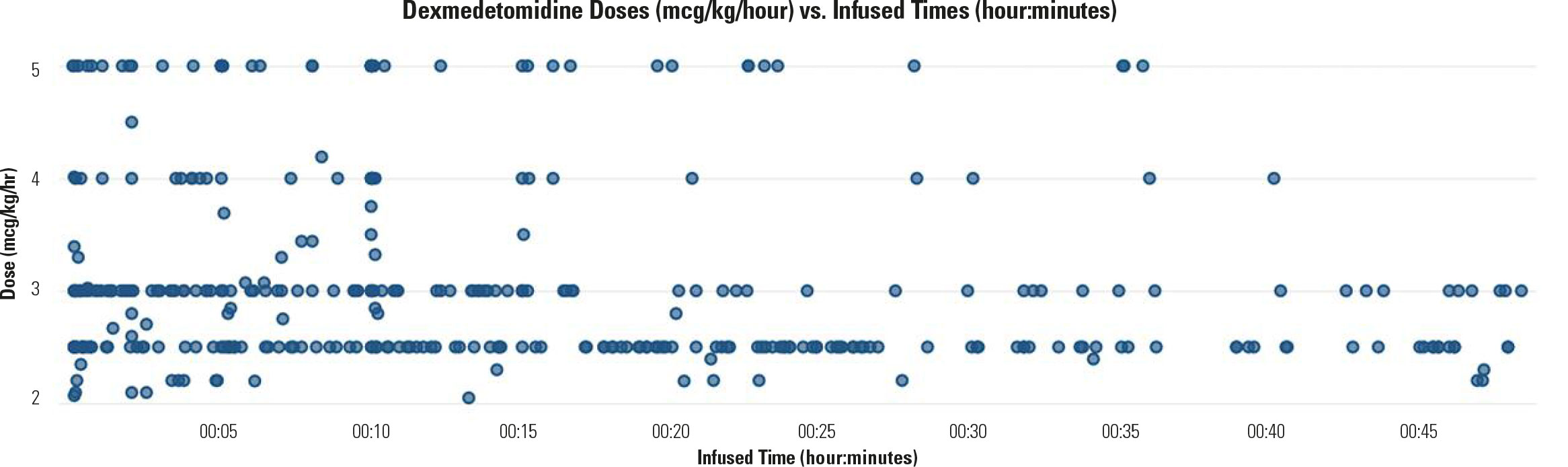
Dexmedetomidine is a sedative often used for procedural sedation. The usual adult dose for a maintenance infusion is 0.2 to 1.5 mcg/kg/hour. Figure 2 shows the distribution of doses administered at or above 2 mcg/kg/hour in 5 network hospitals during the calendar year 2019. The figure shows that, over 12 months, many doses were greater than 2 mcg/kg/hour. Even if the upper dosing limit was 2.5 mcg/kg/hour (although it is thought that doses greater than 1.5 mcg/kg/hour do not add to clinical efficacy),9 quite a few doses exceeded this amount. Dosing was as high as 5 mcg/kg/hour, which infused for as long as 35 minutes in two cases. Dexmedetomidine can cause significant patient harm at high doses, notably severe hypotension and bradycardia.
Learning from the high-dose data
These two examples highlight the value of data that describe the doses commonly administered for OR medications infused via smart pumps. In both examples, there were many programmed doses that were, in some cases, 3- to 4-fold greater than the upper therapeutic range for these medications. The best way to know this is happening is to require infusions to be programmed using smart pumps with DERS, and to have a process in place to evaluate the resulting saved pump data.
To find out why high doses were being administered, these data were brought back to the providers who administered the medications for further investigation. Some of the common practices that were identified as contributing to these high doses included the following:
-
Lack of using a pump’s loading and bolus dose feature. Most of the smart pumps currently on the market offer features that allow clinicians to safely program and administer a loading or bolus dose and then resume the maintenance infusion at the prescribed rate of infusion. This process involves simply entering the desired loading or bolus dose along with the maintenance dose into the pump; once that loading or bolus dose is delivered, the pump begins/resumes administration of the drug at the programmed maintenance infusion rate. Due to a lack of using this loading or bolus feature (in some cases because hard stops had become annoying or staff were not trained to use it), anesthesia providers were often increasing the rate of the maintenance infusion to administer bolus doses. This is a significant safety concern given that there are no prompts or reminders to anesthesia providers to return to a maintenance dose infusion rate once the bolus has been administered (as likely happened with the longer high-dose infusions). Thus, the risk of an overdose is high.
-
Lack of hard stops in anesthesia/OR libraries. Because there were no hard stops for propofol and dexmedetomidine doses that exceeded safe limits, it was easy to bypass any soft stops and alerts that were issued. There are two drivers for lack of hard stops in anesthesia/OR drug libraries. The first involves omission of hard stops when building drug libraries, particularly if there is resistance to hard stops by anesthesia providers. The second involves functionality with some smart infusion pumps that turns alerting hard stops into soft stops when the pump is being used by anesthesia (“anesthesia mode”).
Recommendations
The first hurdle is to ensure anesthesia providers and other OR practitioners appropriately employ DERS when using smart infusion pumps for all medications, including loading and bolus doses, and hydrating fluids. Administering high-alert medication infusions using smart pump technology was one of the key recommendations stemming from a 2010 Anesthesia Patient Safety Foundation (APSF)-sponsored conference for improving medication safety in the OR that focused on Standardization, Technology, Pharmacy/Prefilled/Premixed (Medications), and Culture (STPC).10 The updated ISMP Guidelines for Optimizing Safe Implementation and Use of Smart Infusion Pumps significantly expands expected use to all medications and fluid infusions.11 The following are key to implementing these recommendations and reducing the risk of inappropriate high-dose drug administration in the OR:
Engage anesthesia providers/OR practitioners. Build the anesthesia/OR drug library using an interdisciplinary team that includes anesthesia providers and other OR practitioners. Using this team, implement a systematic process for quarterly review of the library content that includes recent literature, evolving practice changes, facility-specific requests, formulary changes, and pump data. Communicate any changes to the anesthesia/OR drug library to end users.
Implement hard stops in the DERS. Implement upper and lower hard limits for medication doses, concentrations, infusion rates, and loading and bolus doses (often different than hard limits for maintenance infusions) used in the OR. Consider limiting the use of smart pumps in “anesthesia mode,” if available. The use of hard stops can serve as a forcing function and dramatically reduce the incidence of incorrect infusion pump programming.
Make use of DERS an expectation. Leadership needs to establish that the use of DERS in smart infusion pumps is an expected practice in the OR and other perioperative areas for all continuous medication infusions, intermittent and secondary infusions, loading and bolus doses, patient-controlled analgesia infusions, epidural and nerve block infusions, and hydrating solution infusions (except when the hydrating fluid administration rate is greater than the pump allows).
Default to DERS. When possible, use smart pumps that default to programming in DERS and also make it prominently visible when operating outside of DERS.
Require use of the bolus feature. If administering a loading or bolus dose from a continuous medication infusion, only use the bolus feature (with hard limits) that automatically switches to the maintenance infusion rate once the loading or bolus dose has been delivered. Do not allow the delivery of loading or bolus doses from the continuous infusion by simply increasing the rate of infusion.
Provide training. Implement standardized training and competency assessments for the use of smart infusion pumps with DERS for anesthesia providers and other OR practitioners. As stated above, some of the causes that have been identified for programming high doses in the anesthesia care profile involve a lack of understanding of the capabilities of smart pumps. Providing appropriate “hands on” education to help anesthesia providers and other OR practitioners understand the capabilities and potential limitations of smart infusion pumps could help mitigate some unsafe practices, such as administering loading or bolus doses by increasing the continuous infusion rate.
Standardize concentrations. Standardize and limit the number of different drug concentrations and dosing units between the OR and critical care areas for typical infusions started in the OR and likely continued in critical care units. Use pharmacy-prepared or premixed infusions, and ensure the concentrations and dosing units are consistent within the electronic health record and pharmacy IV workflow systems.
Establish transfer protocols. When transferring patients receiving infusions from the OR to a recovery and/or patient care unit, establish a process to ensure the drug library profile is appropriate for the receiving area/unit. If not, establish a procedure to safely switch the patient’s smart pump to the appropriate library and programming.
Use data to understand practice. Provide dedicated time and resources for regular (e.g., at least quarterly) review and analysis of OR smart infusion pump data. Analyze basic and advanced pump metrics8,11,12 to evaluate DERS compliance (goal of 95% or greater), monitor alerts and actions taken in response to alerts, and investigate potential medication errors and/or trends. Providing visibility of the potential for medication errors can be the first step in ultimately reducing the incidence of adverse events in the OR.
Remove barriers. Understanding the barriers to using smart pumps with DERS in the OR is a vital step. This can be accomplished through focus groups of anesthesia providers and other OR practitioners, surveys, other forms of end-user input, and analysis of basic and advanced pump metrics. If barriers or potentially harmful trends are identified, organization-specific improvement plans (e.g., update the library, address nuisance alerts, modify clinical workflow or procedures, identify opportunities for additional education) should be established to remove or reduce these barriers and reduce potentially harmful behaviors.
Seek external assistance with data analytics. In ISMP’s 2018 survey on pump data analytics, we found that 96% of respondents believed that using data from smart infusion pumps was vital to driving quality improvement. However, only 22% of respondents felt that their organization had the correct resources and skills to capture meaningful and actionable insights from this data.12 If this is the case in your organization, ISMP recommends utilizing external resources, such as data companies (e.g., Bainbridge Health, Regenstrief National Center for Medical Device Informatics [REMEDI]) or infusion pump manufacturers, to assist with data evaluation when needed.
*ISMP thanks Sean O’Neill, PharmD, co-founder and chief clinical officer at Bainbridge Health, for providing data and other content for this article.
References
- Rothschild JM, Keohane CA, Cook EF, et al. A controlled trial of smart infusion pumps to improve medication safety in critically ill patients. Crit Care Med. 2005;33(3):533-40.
- Adachi W, Lodolce AE. Use of failure mode and effects analysis in improving the safety of i.v. drug administration. Am J Health Syst Pharm. 2005;62(9):917-20.
- Vanderveen T. Intravenous infusion medication safety: the vision becomes reality. Patient Safety & Quality Healthcare. 2013;10(2):24-30.
- Fields M, Peterman J. Intravenous medication safety system averts high-risk medication errors and provides actionable data. Nurs Adm Q. 2005;29(1):78-87.
- Ohashi K, Dalleur O, Dykes PC, Bates DW. Benefits and risks of using smart pumps to reduce medication error rates: a systematic review. Drug Saf. 2014;37(12):1011-20.
- Vanderveen T, O’Neill S, Beard JW. How can we tell how “smart” our infusion pumps are? APSF Newsletter. 2020;35(1):21-2.
- Schneider PJ, Pedersen CA, Scheckelhoff DJ. ASHP national survey of pharmacy practice in hospital settings: dispensing and administration—2017. Am J Health Syst Pharm. 2018;75(16):1203–26.
- ISMP. Smart pumps in practice: survey results reveal widespread use, but optimization is challenging. ISMP Medication Safety Alert! 2018;23(7):1-5.
- Venn M, Newman J, Grounds M. A phase II study to evaluate the efficacy of dexmedetomidine for sedation in the medical intensive care unit. Intensive Care Med. 2003;29(2):201-7.
- Eichhorn JH. APSF hosts medication safety conference. APSF Newsletter. 2010;25(1):1–20.
- ISMP. Guidelines for optimizing safe implementation and use of smart infusion pumps. 2020.
- ISMP. Survey results: smart pump data analytics. Pump metrics that should be monitored to improve safety. ISMP Medication Safety Alert! 2018;23(14):1-4.
-
Access this Free Resource
You must be logged in to view and download this document.
